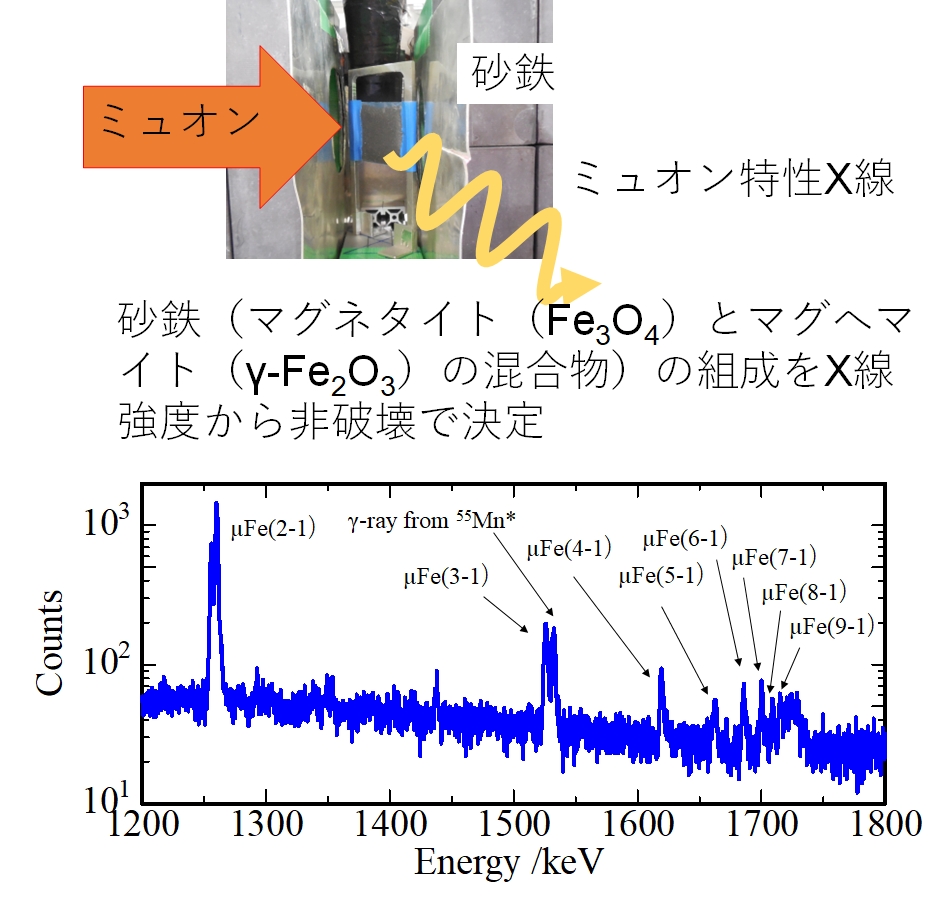
Tuning the Ground- and Excited-State Redox Potentials of Octahedral Hexanuclear Rhenium(III) Complexes by the Combination of Terminal Halide and N-Heteroaromatic Ligands
Takashi Yoshimura,* Hayato Nishizawa, Kojiro Nagata, Akitaka Ito, Eri Sakuda, Shoji Ishizaka, Noboru Kitamura, Atsushi Shinohara
ACS Omega,
2022, 7, 26965 − 26982. DOI:
10.1021/acsomega.2c03834
The present study reports that the ground- and excited-state Re6(23e)/ReRe6(24e) redox potentials of an octahedral hexanuclear rhenium(III) complex can be controlled by systematically changing in the number and type of the N-heteroaromatic ligand (L) and the number of chloride ions at the six terminal positions. Photoirradiation of [Re6(μ3-S)8Cl6]4– with an excess amount of L afforded a mono-L-substituted hexanuclear rhenium(III) complex, [Re6(μ3-S)8Cl5(L)]3– (L = 4-dimethylaminopyridine (dmap), 3,5-lutidine (lut), 4-methylpyridine (mpy), pyridine (py), 4,4ʹ-bipyridine (bpy), 4-cyanopyridine (cpy), and pyrazine (pz)). The bis- and tris-lut-substituted complexes, trans- and cis-[Re6(μ3-S)8Cl4(lut)2]2– and mer-[Re6(μ3-S)8Cl3(lut)3]–, were synthesized by the reaction of [Re6(μ3-S)8Cl6]3– with an excess amount of lut in refluxed N,N-dimethylformamide. The mono-L-substituted complexes showed one-electron redox processes assignable to E1/2[Re6(23e)/Re6(24e)] = 0.49–0.59 V vs. Ag/AgCl. The ground-state oxidation potentials were linearly correlated with the pKa of the N-heteroaromatic ligand [pKa(L)], the 1H NMR chemical shift of the ortho proton on the coordinating ligand, and the Hammett constant (σ) of the pyridyl-ligand substituent. The series of [Re6(μ3-S)8X6–n(L)n]n–4 complexes (n = 0, X = Cl, Br, I, or NCS; n = 1–3, X = Cl) showed a linear correlation with the sum of the Lever electrochemical parameters at the six terminal ligands (ΣEL). The cyclic voltammograms of the mono-L-substituted complexes (L = bpy, cpy, and pz) showed one-electron redox waves assignable to E1/2(L0/L–) = –1.28 to –1.48 V vs. Ag/AgCl. Two types of photoluminescences were observed for the complexes, originating from the cluster core-centered excited triplet state (3CC) for L = dmap, lut, mpy, and py, and from the metal-to-ligand charge-transfer excited triplet state (3MLCT) for L = bpy, cpy, and pz. The complexes with the 3CC character exhibited emission features and photophysical properties similar to those of ordinal hexanuclear rhenium complexes. The emission maximum wavelength of the complexes with 3MLCT shifted to the longer-wavelength in the order L = 4-phenylpyridine (ppy), bpy, cpy, and pz, which agreed with the difference between E1/2[Re6(23e)/Re6(24e)] and E1/2(L0/L–). The calculated oxidation potential of the excited hexanuclear rhenium complex with the 3CC character was linearly correlated with pKa(L), σ, and ΣEL. The ground- and excited-state oxidation potentials were finely tuned by the combination of halide and L ligands at the terminal positions.
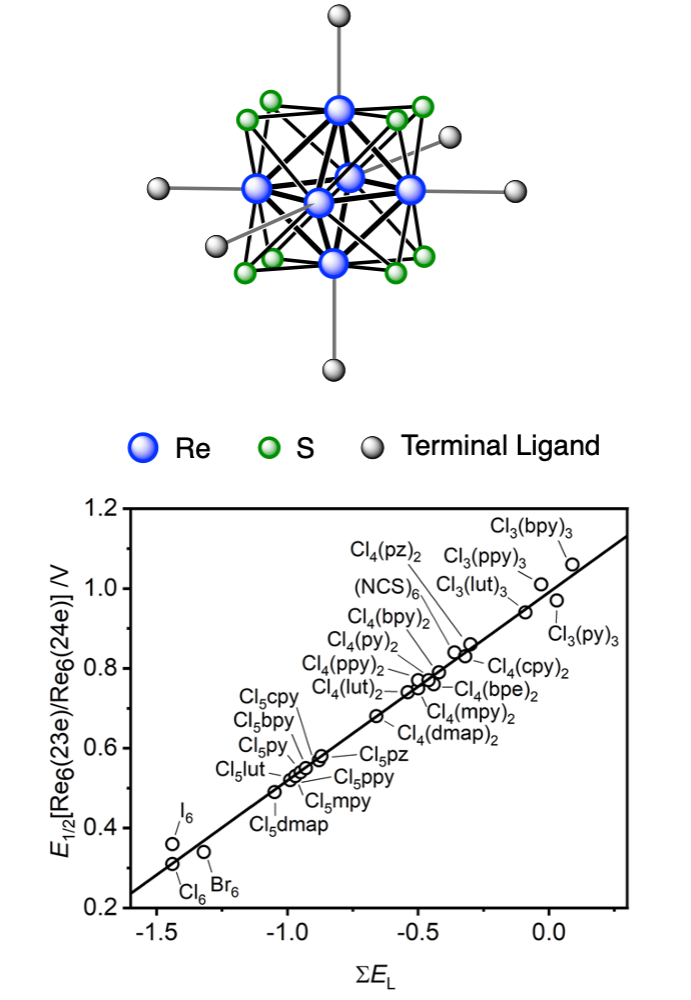
様々な配位子をもつ30種類以上のレニウムの六核錯体の酸化還元データから基底状態の酸化電位と励起状態の酸化電位が電子供与能の指標になる様々なパラメータに対して直線的な相関をもつことを明らかにしました。